Prebiotics And Why You Need Them in Your Diet
Prebiotics are specialized fibers that beneficially nourish the good bacteria already in the large bowel or colon. When prebiotics are used to increase commensal bacterial species such as Bifidobacteria or Lactobacilli toward being the numerically predominant genus in the colon, improved colonization resistance results. These probiotic species are beneficial microorganisms because species within these groups have been reported to exert therapeutic and prophylactic influences on human health...

Prebiotics are specialized fibers that beneficially nourish the good bacteria already in the large bowel or colon. When prebiotics are used to increase commensal bacterial species such as Bifidobacteria or Lactobacilli toward being the numerically predominant genus in the colon, improved colonization resistance results. These probiotic species are beneficial microorganisms because species within these groups have been reported to exert therapeutic and prophylactic influences on human health [1]. In this way, prebiotic compounds are able to modulate both the luminal and mucosal microbial composition and activities, and beneficially regulate host-microbe interactions. Moreover, these changes in the gut microbiota composition (especially the number of Bifidobacteria), contribute to the modulation of metabolic processes associated with common metabolic conditions, such as cardiovascular disease, obesity, and type 2 diabetes [2]. Or, in the simplest terms possible, a prebiotic is a "selectively fermented ingredient that allows specific changes, both in the composition and/or activity in the gastrointestinal microflora that confers benefits upon host well-being and health” [3]. The three criteria of prebiotics are that they must be: (a) Resistant to gastric acids, hydrolysis by mammalian enzymes, and gastrointestinal absorption; (b) Fermented by large intestinal microflora, and; (c) Selective stimulation of the growth and/or activity of intestinal bacteria associated with health and well-being.
Prebiotics
Current Prebiotic Science
“There is no class of biological compounds whose importance is recognised less than the carbohydrates.” Christopher Bucke, University of Westminster The concept of prebiotics has attracted much attention, stimulating scientific as well as commercial interest— and for a good reason. However, of note, some prebiotics receive less interest than they deserve, and are often maligned in health communities. While studies exploring the benefits of dietary fructooligosaccharides (FOS) and galactooligosaccharides (GOS) dominate the scientific literature in the field of prebiotics, a variety of other carbohydrates also show unique promise. These include a wide range of carbohydrate structures from small sugars such as lactulose, as well as various other oligosaccharides, polysaccharides, and resistant starches.
Types of Prebiotics
The non-digestible carbohydrates may be broken down into groups, and include:
- Resistant Starch
- Non-Digestible Oligosaccharides
- Non-Digestible Polysaccharides
Resistant Starch:
As the name suggests, resistant starch is an insoluble starch that is resistant to hydrolysis. They are lower in molecular weight and are relatively short-chain carbohydrates. Resistant starch is selectively fermented by a wide range of colonic bacterial species including members of the Bacteroides spp., Eubacterium spp. Bifidobacteria, and lactobacilli [4] [5]. Natural Sources(Percent of oligosaccharides in total fresh content) [10] [11] : Types of Resistant starch: There are four main types of resistant starch which vary in structure and source. RS1 refers to starch that is physically encapsulated in food, for example, in a fiber mesh or thick cell wall that is unavailable to enzymes. RS2 is a naturally resistant starch due to its crystallinity or dehydrated nature. RS3 is derived from heating and cooling of gelatinized starch. RS4 has been chemically modified, which may include the formation of cross-linkages and esterification [4] [5]. Natural Sources of RS: (grams of RS per 100g of food) [6]: • Oats, Rolled Uncooked 11.3 **•**Puffed wheat 6.2 • Pumpernickel bread 4.5 • Rice Square Cereal 4.3 • Bananas, Raw 4.0 • Italian bread, toasted 3.8 • Potato Chips 3.5 • Plantain cooked 3.5 • Cornflakes 3.2 • Rye bread, wholemeal 3.2 • Tortillas, corn 3.0 • Pizza dough baked 2.8 • Breadsticks, hard 2.3
**Discussion:**The top sources of resistant starch in the American diet are breads, cereals, pastas, and non-leguminous vegetables. These contribute to 21%, 19%, and 19% of the total resistant starch intake, respectively. High levels of resistant starch are naturally found in uncooked rolled oats (7-14% of the total content), cooked and cooled potatoes (19% of the total content). However, more processed forms of resistant starch, like potato starch and corn starch, equate to roughly 66-80% of the material content [6].
Intakes of as little as 6 to 12 grams of resistant starch per meal have been observed to have beneficial effects. These include improving the glycemic control in diabetes, reductions in postprandial glucose and insulin levels, and a potential decrease in the risk for the development of diabetes [7].
However, studies indicate that Americans aged 1 year and older were estimated to consume approximately 4.9 g resistant starch per day when the recommended intake for RS is between 30-50g per day [6]. Americans intakes of RS are considerably low, and efforts should be made to consume more RS-rich foods. Alternatively, scientists have proposed that commercial goods fortified with resistant starch and other forms of prebiotics as a “functional food ingredient” offer a unique solution to help more Americans get the RS they need to see the associated benefits [8].
Prebiotic Oligosaccharides
Lower molecular weight oligosaccharides are relatively short-chain carbohydrates that occur widely in nature. They are typically found in plants but have also been found in human milk and the colostrum of various animals in smaller quantities.
Common Types of Oligosaccharides:
The main types of non-digestible oligosaccharides are fructooligosaccharides (FOS), galactooligosaccharides (GOS), xylooligosaccharides (XOS), isomaltooligosaccharides (IMO) and lactulose. Culture fermentations with human fecal bacteria have shown that FOS, GOS, XOS, IMO, and lactulose alter the microflora, increasing the level of bifidobacteria and/or lactobacilli, causing harmful clostridia and Bacteroides to decline [8]. These low molecular weight compounds are able to withstand digestive processes before they reach the colon, imparting a prebiotic effect. They have been found to effectively stimulate the growth of a limited number of bacteria, leading to a change in the overall microbial balance in the colon [9]. Oligosaccharide Characteristics and Differences [9] : • Xylooligosaccharides and lactulose produce the highest number of bifidobacteria. • Fructooligosaccharides produce the highest populations of lactobacilli. • Galactooligosaccharides resulted in the greatest decrease in clostridia. • Short Chain Fatty Acids (SCFAs) were produced the most by lactulose and GOS. • Gas was produced the most by Inulin. • Isomaltosaccharides and GOS increased bifidobacteria, producing the least gas Natural Sources (Percent of oligosaccharides in total fresh content) [10] [11] :
• Chicory roots 15-24 • Jerusalem artichoke 16-22 • Dandelion 12-15 • Dahlia 13 • Globe Artichoke 3-10 • Salsify 4-11 • Onions 1.1 - 7.5 • Burdock 3.6 • Garlic 1-16 • Leek 2-10 • Wheat 0.8 - 4.0 • Rye 0.5 - 1.0 • Banana 0.3 - 0.7
Discussion:
Results from various studies support the potential of oligosaccharide prebiotics as a means to combat metabolic disorders and intestinal disorders [12]. They also provide evidence that supports the inclusion of supplementation through diet, and commercial products. Not only are oligosaccharides metabolically beneficial, but the physiochemical properties of oligosaccharides may also have broad applications in commercial products, as they are 0.4 to 0.6 times as sweet as sucrose, have lower caloric value, and have been successfully and healthfully added as fortifications in human and animal experimental diets [13].
Non-Digestible Polysaccharides (NDP)
Nondigestible polysaccharides are long, polymeric carbohydrate chains that contain up to several hundred thousand monomers. Each polysaccharide differs by types of monomeric units linked, types of linkages, the order of monomers in the chains, branch points in the molecular backbone, and presence of acid groups. The consumption of NDP has been associated with fecal bulking, binding to other compounds, and the associated benefits of fermentation. These include attenuating blood glucose, maintaining gastrointestinal health, positively affecting the bioavailability of calcium and magnesium, and improving immune function. All the while, improving the rheological properties and nutritional value of foods [14]. Common Sources of Non-Digestible Polysaccharides [15]: • Cellulose • Hemicellulose • Polydextrose • Beta-Glucans • Pectins • Mucilages • Galactomannans • Glucomannans • Tree Resins • Leguminous Gums • Bacterial exopolysaccharides • Seaweed polysaccharides • Chitin, chitosan While these non-digestible polysaccharide fibers do resist hydrolysis and gastrointestinal absorption and are fermented by intestinal flora, it is still essential that the fibers show that they are able to selectively stimulate the growth of commensal bacteria to improve the health and well-being of the host, to be considered as a prebiotic. While it has been found that these non-digestible polysaccharides do have prebiotic effects, the benefits they exert are remarkably different.
Role of Molecular Weight in Prebiotic Potential
The adult human gastrointestinal tract (GIT) is 9 meters or 29.5 feet from the esophagus to the anus. It is important to note that short-chain, low molecular weight monosaccharides and disaccharides are more easily fermented proximally in the gastrointestinal tract than their more resistant and complex, higher molecular weight, oligosaccharide or polysaccharide counterparts [16].
While short-chain prebiotics has shown to impart benefits, the large, slowly fermented polysaccharides of higher molecular weight may have advantages over small, rapidly fermented sugars such as lactulose, and other non-digestible oligosaccharides. These include the ability to be tolerated at higher doses by consumers with reduced risk of side effects such as intestinal discomfort and flatulence caused by excessive gas formation; mucosal damage from rapid acidification; or the laxative effect of too high concentrations of small sugars in the colon [16].
And perhaps more importantly, high-molecular weight polysaccharides supply a persistent source of fermentable carbohydrate throughout the length of the colon rather than being completely fermented proximally. This fact may be of particular interest in the prevention of certain types of diseases as well as colon cancer, as the distal colon and rectum are significant sites of inflammation and disease in humans [17].
A Novel Approach to Prebiotic Supplementation
We know that the fermentation of refined, and short-chain carbohydrates and oligosaccharides occur more proximally, whereas the more complex oligosaccharides and polysaccharides can be fermented distally [16]. We also know that a highly refined “Western Diet” high in saturated fat while lacking in complex carbohydrates is associated with several metabolic, and autoimmune diseases [18]. Carbohydrate complexity is associated with molecular weight. The lower the molecular weight, the shorter the chains. The higher the weight, the higher the number of linkages. However, the most complex polysaccharides with the highest molecular weight that are most often found in the diet are food-grade hydrocolloids. Figure 2: Viscosity and Molecular Weight Correlation. Adapted from Reference 19.
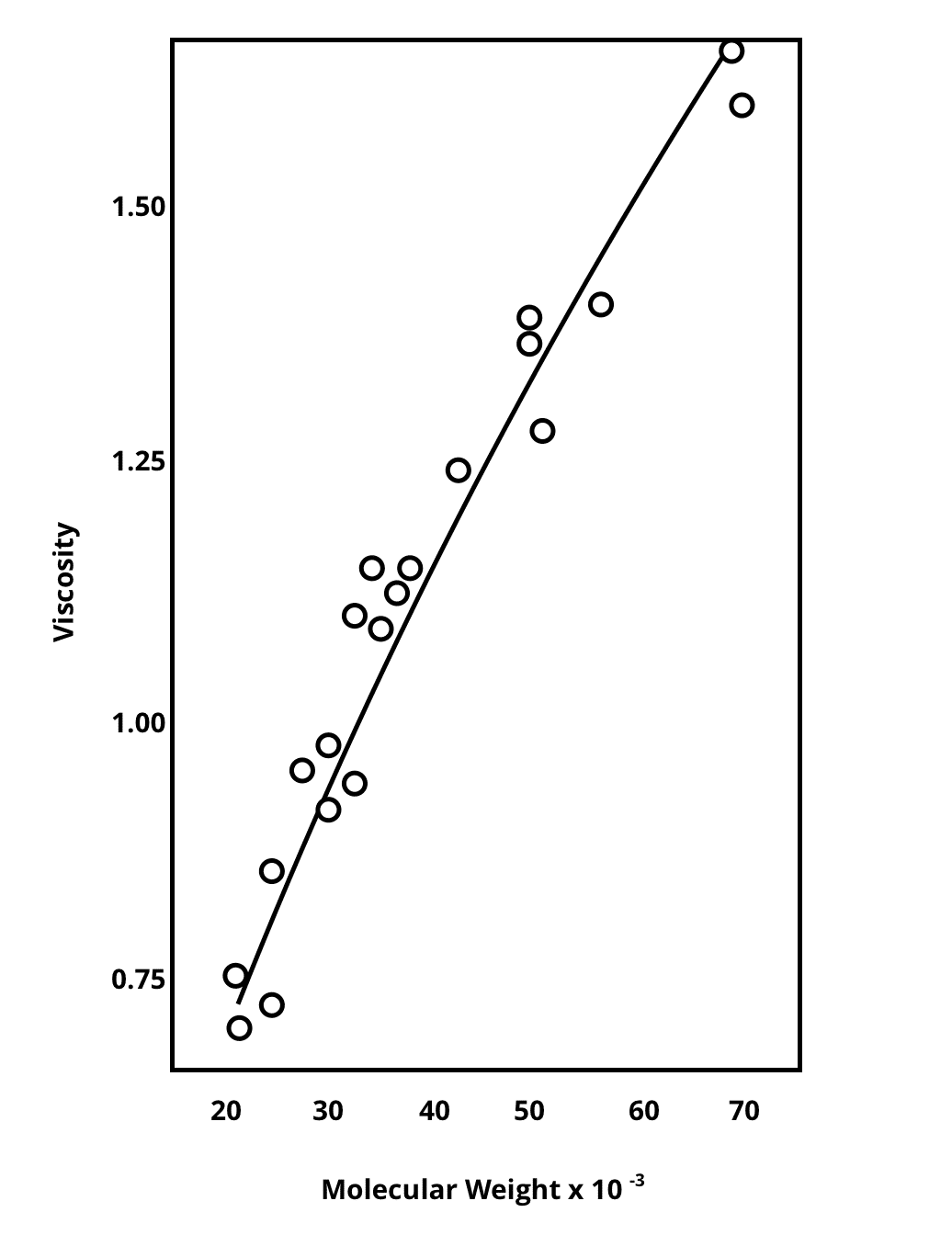
The relationship between the molecular weight and complexity of an ingredient is highly correlated with its viscosity [19, Figure 2] High molecular weight is an attractive feature of commercial hydrocolloids, or substances that form a gel in the presence of water. High viscosity, high-molecular-weight, non-digestible, complex polysaccharides include pectins, arabinogalactans, b-glucans, inulin, mucilages, as well as bacterial gums like xanthan gum or gellan gum; tree gums, such as gum arabic, gum tragacanth, and gum ghatti; leguminous gums, such as guar gum and locust bean gum; as well as food-grade, water-soluble seaweed extracts, such as carrageenan, sodium alginate, and agar [15]. These ingredients aren’t just dietary fibers, they are complex polysaccharides that have shown to impart prebiotic effects when consumed.
Discussion
Despite the healthful influence prebiotic fiber can have on reducing the risk of chronic disease, the intake remains low in many populations worldwide, in particular in the U.S.A. Amount of whole foods needed to meet 30-50 grams suggested prebiotic intake per day [20] [21]:
| Whole Food | Percent of Total Prebiotic Fiber | Daily Recommended Intake |
| Raw Chicory Root | 65 | 55.8 - 93.0 grams /day 1.97 - 3.28 oz / day |
| Raw Jerusalem Artichoke | 31.5 | 114 - 190 grams/ day 4.02 - 6.70 oz / day |
| Raw Dandelion Greens | 24.3 | 148.2 - 247.0 g/day 5.23 - 8.71 oz/ day |
| Raw Garlic | 17.5 | 205.8 - 343.0 g/ day 7.23 - 12.10 oz/ day |
| Raw Leeks | 11.7 | 307.8 - 513.0 grams/day 10.86 - 18.10 oz/day |
| Raw Onion | 8.6 | 418.8 - 698.0 grams/day 14.77- 24.62 oz |
| Cooked Onions | 5 | 720-1200 grams/day 25.4 - 43.33 oz/day |
| Raw Wheat Bran | 5 | 720-1200 grams/day 25.4 - 43.33 oz/day |
| Baked Wheat Flour | 4.8 | 3600 - 6000 g/day 126.99 - 211.64oz |
Based on this information, it should be evident that it would be extremely difficult to get 30-50 grams of fiber per day with commonly available foods. Amount of hydrocolloids and processed starch needed to meet 30-50 grams suggested prebiotic intake per day:
| Prebiotic Fiber | Percent of Total Prebiotic Fiber | Daily Recommended Intake |
| Hi-Maize Corn Starch | 50 | 60-100 grams/day 2.1 - 2.2 oz/day |
| Potato Starch | 75 | 40-67 grams/day 1.41 - 2.36 oz/day |
| Gum Arabic | 98.5 | 31-51 grams/day 1.09 - 1.8oz/day |
| Xanthan Gum | 100 | 30-50 grams/day 1.05 - 1.07 oz/day |
| Isolated Inulin | 100 | 30-50 grams/day 1.05 - 1.07 oz/day |
| Guar Gum | 100 | 30-50 grams/day 1.05 - 1.07 oz/day |
Getting adequate complex carbohydrate intake can be a challenge. Some suggest that even 30-50 grams of prebiotic fiber are too low, as some studies conclude that the average daily intake of prebiotic fiber for our hunter-gatherer ancestors may have been over 100 grams per day. Nevertheless, the grossly overlooked and wholly underappreciated hydrocolloids and food starches present a unique solution to this problem, as foods may be fortified with prebiotics of low and high molecular weight to improve the nutritive value of foods. For this reason, we propose that the aforementioned prebiotics, including food gums especially, are considered healthy, functional ingredients by those in the food industry and health industry as well.
References
- Vernazza, C.L., Rabiu, B.A. Gibson, G. R. (2006) Human Colonic Microbiology and the Role of Dietary Intervention: Introduction to Prebiotics. In: Prebiotics Development & Application. G.R. Gibson & R.A. Rastall (Ed.) Hoboken, New Jersey: John Wiley & Sons. (pp 11-21)
- Yoo, J., & Kim, S. (2016). Probiotics and Prebiotics: Present Status and Future Perspectives on Metabolic Disorders. Nutrients, 8(3), 173. doi:10.3390/nu8030173
- Yoo, J., & Kim, S. (2016). Probiotics and Prebiotics: Present Status and Future Perspectives on Metabolic Disorders. Nutrients, 8(3), 173. doi:10.3390/nu8030173
- Englyst, H. N., & Cummings, J. H. (1986). Digestion of the carbohydrates of banana (Musa paradisiaca sapientum) in the human small intestine. The American Journal of Clinical Nutrition, 44(1), 42–50. doi:10.1093/ajcn/44.1.42
- Cummings, J. H., Beatty, E. R., Kingman, S. M., Bingham, S. A., & Englyst, H. N. (1996). Digestion and physiological properties of resistant starch in the human large bowel. British Journal of Nutrition, 75(05), 733. doi:10.1079/bjn19960177
- Murphy, M. M., Douglass, J. S., & Birkett, A. (2008). Resistant Starch Intakes in the United States. Journal of the American Dietetic Association, 108(1), 67–78. doi:10.1016/j.jada.2007.10.012
- Behall, K., & Hallfrisch, J. (2002). Plasma glucose and insulin reduction after consumption of breads varying in amylose content. European Journal of Clinical Nutrition, 56(9), 913–920. doi:10.1038/sj.ejcn.1601411
- Roberfroid, M. (2002). Functional food concept and its application to prebiotics. Digestive and Liver Disease, 34, S105–S110. doi:10.1016/s1590-8658(02)80176-1
- Rycroft, C. E., Jones, M. R., Gibson, G. R., & Rastall, R. A. (2001). A comparative in vitro evaluation of the fermentation properties of prebiotic oligosaccharides. Journal of Applied Microbiology, 91(5), 878–887. doi:10.1046/j.1365-2672.2001.01446.x
- Modler, H. W. (1994). Bifidogenic factors—sources, metabolism and applications. International Dairy Journal, 4(5), 383–407. doi:10.1016/0958-6946(94)90055-8
- Roberfroid, M., Gibson, G. R., & Delzenne, N. (2009). The Biochemistry of Oligofructose, a Nondigestible Fiber: An Approach to Calculate Its Caloric Value. Nutrition Reviews, 51(5), 137–146. doi:10.1111/j.1753-4887.1993.tb03090.x 12.Yasmin, A., Butt, M. S., Afzaal, M., van Baak, M., Nadeem, M. T., & Shahid, M. Z. (2015). Prebiotics, gut microbiota and metabolic risks: Unveiling the relationship. Journal of Functional Foods, 17, 189–201. doi:10.1016/j.jff.2015.05.004
- L’homme, C., Peschet, J. ., Puigserver, A., & Biagini, A. (2001). Evaluation of fructans in various fresh and stewed fruits by high-performance anion-exchange chromatography with pulsed amperometric detection. Journal of Chromatography A, 920(1-2), 291–297. doi:10.1016/s0021-9673(00)01262-0
- Tungland, B. C., & Meyer, D. (2002). Nondigestible Oligo- and Polysaccharides (Dietary Fiber): Their Physiology and Role in Human Health and Food. Comprehensive Reviews in Food Science and Food Safety, 1(3), 90–109. doi:10.1111/j.1541-4337.2002.tb00009.x
- Lewis, B. A., Hall, M. B. and Van Soest, P. J. (2001) Interaction between human gut bacteria and dietary fiber substrates. In: CRC Handbook of Dietary Fiber in Human Nutrition, 3rd Edition. G. A. Spiller (Ed.). CRC Press, Boca Raton, FL, pp. 271–276.
- Crittenden, Ross. (2006) Emerging Prebiotic Carbohydrates. In: Prebiotics Development & Application. G.R. Gibson & R.A. Rastall (Ed.) Hoboken, New Jersey: John Wiley & Sons. (pp 120)
- Crittenden, Ross. (2006) Emerging Prebiotic Carbohydrates. In: Prebiotics Development & Application. G.R. Gibson & R.A. Rastall (Ed.) Hoboken, New Jersey: John Wiley & Sons. (pp 126-160)
- Manzel, A., Muller, D. N., Hafler, D. A., Erdman, S. E., Linker, R. A., & Kleinewietfeld, M. (2013). Role of “Western Diet” in Inflammatory Autoimmune Diseases. Current Allergy and Asthma Reports, 14(1). doi:10.1007/s11882-013-0404-6]
- Mortimer, G. A., Daues, G. W., & Hamner, W. F. (1964). Relationships between molecular weight, solution viscosity, and melt index for narrow distribution, high-pressure polyethylene whole polymers. Fast determinations. Journal of Applied Polymer Science, 8(2), 839–847. doi:10.1002/app.1964.070080223
- Roberfroid, M., Gibson, G. R., & Delzenne, N. (2009). The Biochemistry of Oligofructose, a Nondigestible Fiber: An Approach to Calculate Its Caloric Value. Nutrition Reviews, 51(5), 137–146. doi:10.1111/j.1753-4887.1993.tb03090.x 21. Modler, H. W. (1994). Bifidogenic factors—sources, metabolism and applications. International Dairy Journal, 4(5), 383–407. doi:10.1016/0958-6946(94)90055-8
Related Reading
- Why You Should Be Honest With Your Health Coach
- Why You Need a Paleo Food Broker?
- Coprolites Reveal Prebiotics in Pre-Agricultural Human Diet
Share:



